Comprehensive
Cell Solutions
From Cell Sourcing to GMP Manufacturing:
Your Partner in Cell & Gene Therapies
Comprehensive Cell Solutions (CCS) provides outsourced services for cell and gene therapies through a differentiated enterprise model that integrates scientific expertise with operational excellence. Leveraging over two decades of experience and the advanced infrastructure of New York Blood Center Enterprises, CCS delivers critical cell sourcing, development, and manufacturing solutions to support early-stage to fully integrated biotherapeutic developers. Each engagement with CCS drives scalable innovation while sustaining essential healthcare services and research that impact millions globally.
Draw-to-ThawTM
Platform
Cell Sourcing & Apheresis
Process Development
Analytical Development
cGMP & GTP MANUFACTURING
Cryopreservation
Storage & Logistics
Quality Control & Regulatory
Featured
News

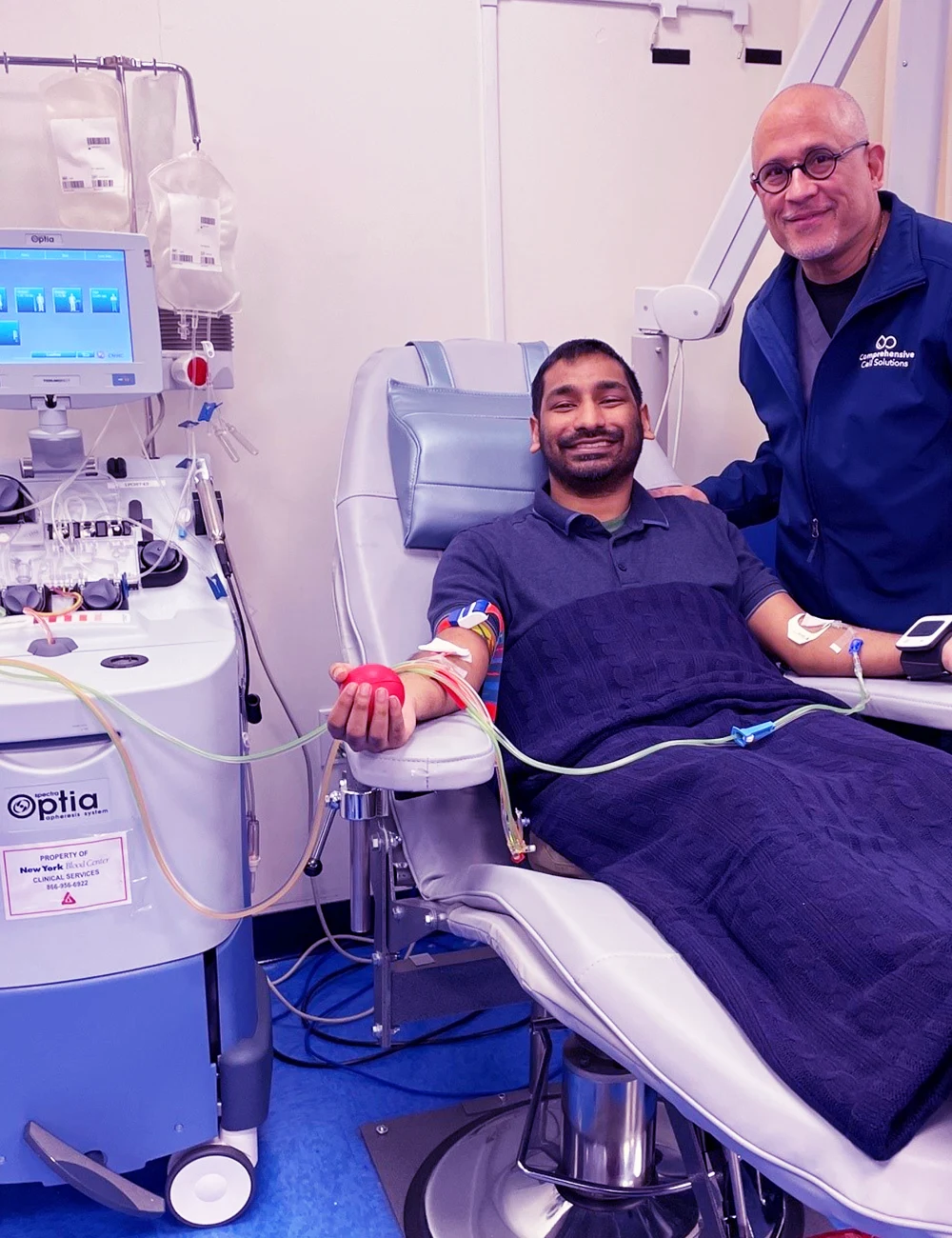
CCS Celebrates Apheresis Awareness Day
In recognition of Apheresis Awareness Day on September 16, Comprehensive Cell Solutions hosted an event to celebrate our apheresis team.
New York Blood Center Enterprises and Human Life CORD Sign Letter of Intent to Expand Manufacturing Collaboration for Umbilical Cord-Derived Mesenchymal Stromal Cell
Exciting partnership aims to drive innovation and enhance global supply chain for next-generation umbilical cord tissue-derived MSC therapies.
Events
Advanced Therapies Week (ATW)
February 9th-12th, 2026 | Location: San Diego CA
BCA Advanced Therapies Workshop
February 17-19, 2026 | Fort Myers, FL
New Jersey Inaugural Life Science Symposium
March 23, 2026 | Piscataway NJ
American Society for Apheresis
April 9-11, 2026 | Montreal Canada
ISCT
May 6-9, 2026 | Dublin Ireland
ASGCT
May 11-15, 2026 | Boston MA
Bio International
June 22-25, 2026 | San Diego CA
Let’s Talk about Solutions!
Advancing Innovation in Cell and Gene Therapies
Partner with CCS for process or analytical development, streamline manufacturing, and optimize cell therapy workflows. We will help you accelerate any advanced therapy with GMP-compliant solutions designed for scalability.
Contact Us
Explore how our portfolio of Draw-to-Thaw™ services, including apheresis expertise phase appropriate development strategies, and capacity for GMP manufacturing can help you achieve your most critical goals.
Let’s collaborate to bring transformative therapies to patients – faster and more affordably.